
Table of Contents
1. Introduction
On February 2, 2026, the U.S. Food and Drug Administration implemented the Quality Management System Regulation (QMSR), replacing the legacy Quality System Regulation (QSR) under 21 CFR Part 820.
By incorporating ISO 13485:2016 into federal law and retiring the QSIT inspection framework, the FDA introduced the most significant structural change to U.S. medical device oversight in decades.
The headline is not harmonization.
The headline is this:
Risk management now drives inspections. Audit documentation is no longer shielded. AI lifecycle governance is under regulatory scrutiny.For pharmaceutical, biotech, and AI-enabled medical technology organizations in GxP environments, this fundamentally changes compliance operationalization.
2. QMSR in depth: what actually changed
The most significant transformation under QMSR is philosophical, not structural.
Under the former QSIT framework, FDA inspections focused on subsystems such as CAPA, Design Controls, Production & Process Controls, and Management Controls. Investigators followed structured sampling logic, evaluating compliance through documentation presence and adequacy.
Under QMSR, that paradigm shifted.
Inspections now organize around six broader QMS Areas, sampling tables are eliminated, and investigator discretion expanded. Most importantly, risk documentation determines inspection depth and direction (@ Allyson B. Mullen & @ Lisa M. Baumhardt ., FDA Law Blog, Understanding FDA’s Risk-Based Inspection Model Under QMSR).
Investigators are instructed to:
Use risk files to prioritize review areas
Trace risk-based decisions across design, supplier management, change control, and postmarket surveillance
Expand inspection scope if risk integration appears weak or fragmented
Evaluate whether executive and management decisions demonstrate true risk-based governance
This shifts focus from documentation sufficiency to governance integrity.
The new Compliance Program Manual explicitly identifies risk management failures as grounds for Official Action Indicated (OAI) classifications, including:
Failure to integrate postmarket surveillance data into risk management files
Failure to evaluate design or software changes for risk impact
Inadequate data analysis resulting in unmitigated health consequences
Weak or superficial change control impact assessments
Risk management is no longer a static regulatory artifact maintained for audit readiness; it is a dynamic enforcement lever.
Under QMSR, the FDA assesses whether risk-based decisions are integrated, traceable, and actively govern the organization, not just whether a quality system exists.

3. The elimination of audit documentation protection
The most disruptive change under QMSR is the removal of FDA’s historical policy of not reviewing:
Internal audit reports
Supplier audit reports
Management review documentation
That policy has been eliminated (@ Greg Matson, Preparing for QMSR in 2026).
Investigators may now request these records, including historical documentation generated before February 2, 2026.
What this means in practice
Many organizations historically treated internal audits as periodic exercises with narrative summaries that varied in quality across auditors and lacked standardization among suppliers.
Under QMSR, those audit records can now be scrutinized for
Trend blindness
Inconsistent findings
Superficial assessments
Disconnects between audit findings and CAPA
Management inaction despite recurring signals
The audit process itself has become inspectable risk.
This is where cIGA becomes strategically critical.

4. How cIGA supports the new audit reality
xLM’s Continuous Intelligent GxP Audit (cIGA) solution was designed for this compliance environment.
cIGA transforms audit execution from episodic documentation into structured, AI-orchestrated intelligence.
What cIGA enables:
AI-guided, standardized audit questioning aligned to GxP controls
Dynamic follow-up based on responses
Real-time validation of evidence
Structured documentation capture
Automated compliance scoring
Complete audit-ready reporting with traceability
In a QMSR environment where audit documentation is reviewable, cIGA delivers:
Consistency across auditors - Eliminates variability in question framing, interpretation, and documentation.
Structured evidence capture - Ensures auditee responses and artifacts are traceable and organized.
Early risk visibility - AI-assisted scoring identifies emerging risk signals before escalation.
Audit scalability - Enables parallel supplier audits without increasing headcount.
Inspection-ready documentation - Produces defensible, standardized reports that withstand regulatory scrutiny.
When audit records become discoverable, audit discipline becomes strategic infrastructure.
cIGA does not replace auditors; it amplifies, standardizes, and makes their work inspection-ready by design (@xLM Continuous Intelligence, AI in GxP Manufacturing, Reengineering Vendor Audits for the AI Era).

5. AI systems under QMSR: validation must be continuous
QMSR’s elevation of lifecycle risk management is especially consequential for AI-enabled systems.
AI introduces dynamic risk variables:
Algorithmic drift
Model retraining cycles
Data distribution shifts
Bias evolution
Adversarial manipulation risk
Continuous software updates
Under QMSR:
Every model update is a change control event
Postmarket performance must feed into risk management
Validation must trace to risk controls
Management must demonstrate oversight of AI risk
One-time validation is insufficient. AI requires continuous validation.

6. How cIV operationalizes AI risk governance
xLM’s Continuous Intelligent Validation (cIV) platform embeds validation into the AI lifecycle.
cIV enables:
AI-assisted generation of User Requirements Specifications (URS)
Automated generation of test cases aligned to risk controls
Two-way traceability matrices
Automated test execution and evidence capture
Structured logs, screenshots, and validation artifacts
Continuous re-validation following AI model updates
In a QMSR context, cIV supports:
Drift validation - Testing model performance against predefined thresholds after retraining.
Bias monitoring - Validating outputs across demographic or use-case variations.
Change impact analysis - Documenting how updates affect risk controls.
PCCP support - Operationalizing Predetermined Change Control Plans through controlled validation workflows.
Audit-ready documentation - Maintaining structured validation evidence for FDA inspection or Remote Regulatory Assessment (RRA).
Where QMSR requires lifecycle risk governance, cIV provides the execution layer (@xLM Continuous Intelligence, AI in GxP Manufacturing, Continuous Intelligent Validation (cIV): From Months of Manual Validation to Minutes of Intelligent Execution).

7. The bigger signal: from compliance to continuous governance
QMSR signals a regulatory evolution:
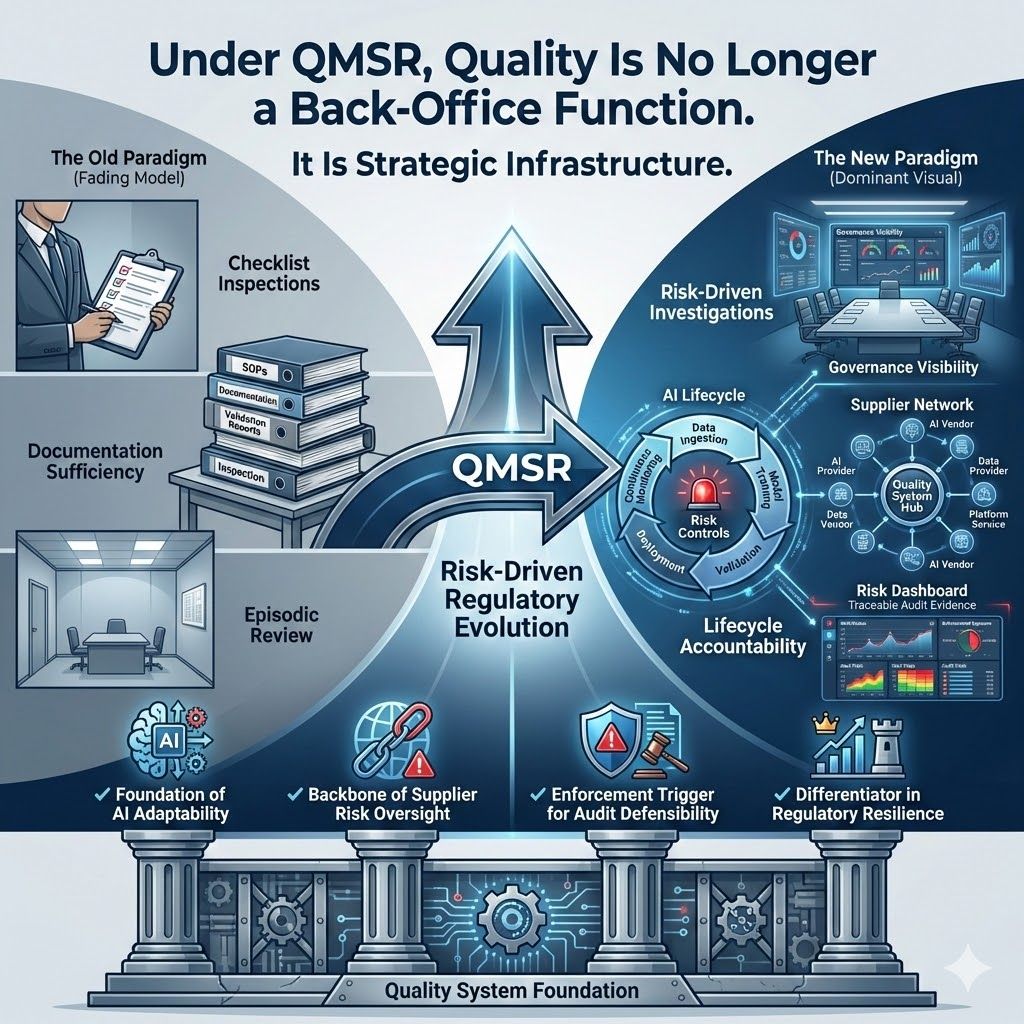
From checklist inspections to risk-driven investigations
From documentation sufficiency to governance integrity
From episodic review to lifecycle accountability
For pharma companies deploying AI-enabled systems and managing complex vendor ecosystems, the quality system is no longer a back-office function.
It is:
The foundation of AI adaptability
The backbone of supplier risk oversight
The enforcement trigger for audit defensibility
The differentiator in regulatory resilience
8. Final Thoughts : cIGA + cIV in a QMSR world
QMSR is not about more paperwork. It is about integrated governance.
cIGA addresses governance at the supplier and internal audit level. cIV addresses governance at the AI model and validation lifecycle level.
Together, they create:
Structured audit intelligence
Continuous validation traceability
Closed-loop risk integration
Scalable compliance execution
Executive visibility into quality signals
This aligns directly with QMSR’s emphasis on:
Risk-based inspection
Management accountability
Data-driven oversight
Continuous improvement

