
Table of Contents
1. Rethinking API Production in an Era of Resilience and Innovation
The pharmaceutical industry is evolving due to supply chain risks, new regulations, and the need for agility. Traditional batch manufacturing is shifting to continuous processes, digitally enabled factories, and AI-driven quality intelligence, boosting efficiency, quality, and global API supply resilience.
In the U.S., domestic API manufacturing is gaining attention, supported by the FDA’s Emerging Technology Program (ETP). This program promotes innovation through regulatory collaboration, helping companies design facilities with continuous flow chemistry, modular infrastructure, and real-time quality monitoring, setting new pharma production standards.
To discuss this shift, xLM Continuous Intelligence hosts a LinkedIn Live conversation with John Raech, Co-Founder and CEO, Pharma Resource Group (PRG). A former Johnson & Johnson leader, John has extensive experience in product development, manufacturing, and regulatory operations, successfully integrating innovation in regulated settings.
At PRG, John leads the creation of a state-of-the-art API manufacturing facility in Pennsylvania, designed for continuous flow chemistry and advanced manufacturing. The site aims to qualify under the FDA’s ETP, positioning PRG as a leader in resilient, high-assurance U.S. API supply. His leadership blends innovation, compliance, and digital transformation for scalable manufacturing.
This unscripted, presentation-free podcast-style discussion offers candid insights into challenges and opportunities in shifting from batch to continuous, digitally enabled manufacturing systems. John's strategic and operational views provide valuable lessons for modernizing manufacturing while ensuring regulatory compliance.

2. A candid conversation: no slides, no scripts
Unlike traditional webinars, this LinkedIn Live session is an unscripted, presentation-free discussion.
Hosted by Nagesh Nama, the conversation focuses on:
Architecting an FDA-aligned digital factory from day one.
Leveraging the FDA Emerging Technology Program (ETP) to de-risk innovation.
Applying continuous flow and real-time quality in regulated API manufacturing.
Using AI and advanced analytics while maintaining GxP and validation rigor.
Building competitive advantage through speed, quality, and regulatory confidence
This format creates a transparent, engaging discussion based on real experience rather than theory.

3. Building a State-of-the-Art API Facility in the United States
Pharma Resource Group (PRG) is developing a state-of-the-art API manufacturing facility in Pennsylvania. This facility will leverage the latest in continuous flow chemistry and advanced manufacturing. The site aims to qualify under the FDA’s Emerging Technology Program (ETP), positioning PRG as a leader in resilient, high-assurance U.S.-based API supply chains.
This initiative marks a shift from traditional batch manufacturing to a modular, digitally enabled, continuously operated facility. This design enables faster technology transfer, better process control, and improved product quality, ensuring higher reliability and efficiency throughout manufacturing.

4. From Continuous Flow to a Fully Digital Factory
During the session, John will share how PRG is rethinking traditional API production through:
1. Architecting an FDA-Aligned Digital Factory
Designing a facility with regulatory alignment from day one ensures innovation does not compromise compliance. Embedding digital infrastructure and data integrity early streamlines validation and accelerates time to market.
2. Leveraging the FDA Emerging Technology Program (ETP)
The FDA’s ETP offers a collaborative path for companies adopting innovative manufacturing technologies. Participants can reduce implementation risks while ensuring regulatory confidence and smoother approvals.
3. Applying Continuous Flow and Real-Time Quality
Continuous manufacturing and real-time monitoring enable tighter process control, less variability, and better product quality, transforming API design, transfer, and release.
4. Harnessing AI and Advanced Analytics within GxP Frameworks
Advanced analytics and AI/ML-driven process control provide predictive insights and proactive risk management while maintaining GxP compliance and validation.
5. Building Competitive Advantage
Organizations integrating digital intelligence with continuous manufacturing gain speed, quality, supply resilience, and regulatory confidence.

5. xLM’s Role: Enabling Continuous Intelligence
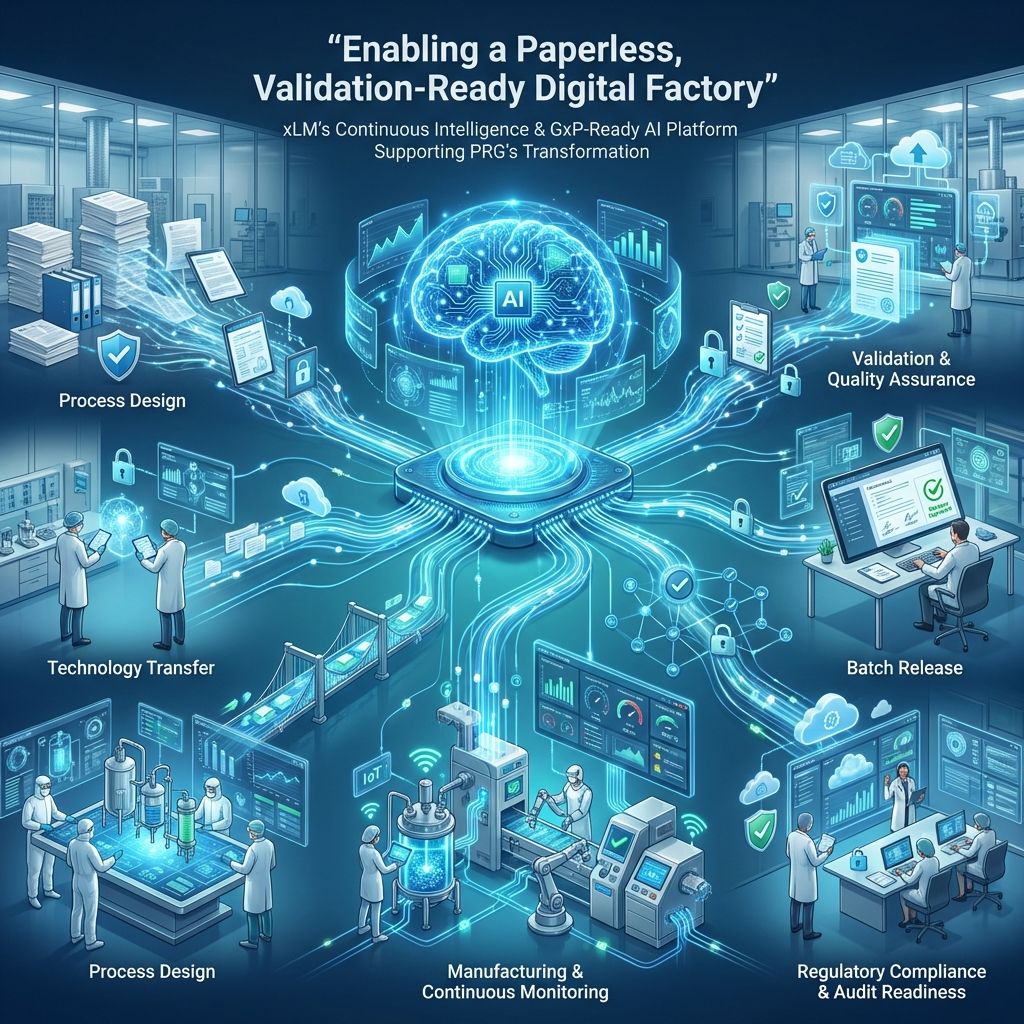
xLM will comprehensively outline how its Continuous Intelligence and GxP-ready AI platform robustly supports PRG’s journey toward achieving a paperless, fully digitized, validation-ready operation. This platform is designed to streamline and enhance every aspect of the digital twin platform, ensuring that compliance is built-in from day 1.
This comprehensive support encompasses every critical phase, starting from process design and technology transfer and extending all the way through to batch release and ongoing regulatory compliance. By leveraging xLM’s advanced solutions, organizations are empowered to effectively operationalize innovation while maintaining full traceability and ensuring audit readiness at all times. This approach not only enhances operational efficiency but also guarantees adherence to stringent regulatory requirements throughout the product lifecycle.
6. What Will You Learn?
This LinkedIn Live session offers actionable insights and strategic perspectives for professionals shaping pharmaceutical manufacturing. By attending, you will:
Understand how to design an FDA-aligned digital factory from the ground up, ensuring compliance while enabling innovation.
Gain practical insights into leveraging the FDA Emerging Technology Program (ETP) to accelerate adoption of advanced manufacturing technologies.
Explore benefits of continuous flow chemistry and real-time quality monitoring in improving efficiency and product assurance.
Learn how AI and advanced analytics can be implemented within GxP environments while maintaining validation and regulatory rigor.
Discover strategies for building competitive advantage through enhanced speed, supply chain resilience, and regulatory confidence.
Hear firsthand experiences from an industry veteran, including lessons learned from transitioning traditional manufacturing paradigms to next-generation approaches.

7. Who Should Attend
This session suits professionals shaping pharmaceutical manufacturing's future and seeking to align innovation with regulatory excellence.
Manufacturing and Operations Leaders
Process Development Scientists
Quality and Compliance Professionals
Digital Transformation and IT Leaders
Regulatory Affairs Strategists
Supply Chain and CMC Executives

8. Join the Live Conversation
The future of pharmaceutical manufacturing lies at the intersection of continuous processes, digital intelligence, and regulatory innovation. Organizations embracing this convergence today will deliver resilient supply chains and superior product quality tomorrow.
If your organization explores next-generation API manufacturing, continuous processing, or integrating digital and AI-driven technologies within regulated environments, this LinkedIn Live event offers practical insights from an industry veteran leading a state-of-the-art API facility in the U.S. Hear how innovation, regulatory alignment, and digital intelligence converge to shape pharmaceutical production's future.
📅 LinkedIn Live Event
April 23, 2026 | 12:00 PM EST
Designing the Future of API Manufacturing: Continuous Flow, Digital Factories, and FDA Emerging Technologies
Featuring
John Raech – Co-Founder & President, Pharma Resource Group (PRG)
Former Johnson & Johnson Executive
Hosted by
Nagesh Nama – CEO, xLM Continuous Intelligence
This unscripted, presentation-free podcast-style discussion is a timely conversation for pharmaceutical leaders seeking to improve manufacturing agility, strengthen supply chain resilience, and implement digital and AI-driven capabilities while maintaining GxP and regulatory rigor.
👉 Reserve your spot to join the live conversation and gain practical lessons from leaders at the forefront of next-generation pharmaceutical manufacturing.

